Normal pH regulation in the kidney
Where does acid normally come from?
A major source of hydrogen ions is the metabolism of sulphur-containing
amino acids, and phosphorylated moieties such as phosphoserine. This is
normally in the order of 50 mEq/day. Production of carbonic acid from
CO2 made during aerobic respiration is not normally a substantial source,
as the CO2 is excreted via the lungs. Other potential sources of acid are
organic acids either produced or ingested. These will be considered when
we discuss acidosis with an elevated anion gap.
How does the kidney get rid of acid (H+ ions)?
The kidney has several mechanisms of acidifying the urine.
Acidification of the urine occurs in both proximal and distal tubules:
- Proximal tubule:
More acid is secreted here than in the distal tubule,
using an antiport mechanism: sodium ions are absorbed from
the lumen of the tubule and hydrogen ions are secreted into the
lumen in exchange. Where do the hydrogen ions come from? From
the dissociation of carbonic acid into HCO3- and
hydrogen ion! The net effect of all this is most beneficial to
the organism - precious sodium and bicarbonate ions are retained,
in exchange for the excretion of unwanted hydrogen ions.
(A tiny amount of hydrogen ion may be secreted into the
proximal tubule by other mechanisms).
- Distal tubule & collecting duct:
The mechanism in the distal tubule is quite different - although
quantitatively smaller, it allows the organism to "fine-tune"
urinary pH. Loss of the mechanism results in difficulty acidifying
the urine substantially. The main method of distal tubular
acidification is using a hydrogen ion pump, driven by ATP. This
pump may be similar to the proton pump in the stomach.
(There is some evidence that there may also be an electroneutral
H+/K+ antiporter working in the distal tubule).
The minimum urinary pH that the kidney can achieve is about 4.5. Although
this is about a thousand times more hydrogen ion than is normally present
in blood (a pH of 7.4 corresponds to a hydrogen ion concentration of
about 4 * 10-8, 4.5 to about 3 * 10-5), as total
quantities go, this is a trivial amount of acid. The kidney needs a better
way of getting rid of hydrogen ions. It has two:
- monobasic phosphate (H2PO4-)
the reaction:
H+ + HPO42-
= H2PO4-
is used to buffer the hydrogen ions, and the monobasic phosphate
is then lost in the urine;
- ammonium ions (NH4+).
The first method is an important mechanism for getting rid of
hydrogen ions (about 10-40 mmol/24 hours, or up to double this if
required), but when the kidney is presented with an extra acid load
above and beyond normal H+ production, the second, ammonium-based
mechanism comes into prominence.
Normally the ammonium mechanism removes 30(+) mmol/24 hours, but this
may increase to 200mmol or more if required. We can measure the buffering
provided by monobasic phosphate quite simply - just add alkali until
the pH of the urine rises to physiological pH (7.4) and this is
a measure of the amount of hydrogen ion buffered.
Unsurprisingly, we call this the "titratable acidity" of the urine.
Measuring the contribution made by ammonium ion is a bit more tricky!
Where does the ammonium ion in the urine come from?
The main source is
removal of ammonia (NH3) from the amino acid glutamine, which is turned
into glutamate. The enzyme glutaminase, abundantly present in renal tubular
cells, catalyses the reaction. The glutamate in turn can give up another
ammonia molecule, to form alpha-ketoglutarate. Ever cunning, the kidney
then can metabolise the alpha-ketoglutarate, using two hydrogen ions in
the process! All in all, this is a smart way of getting rid of ammonia,
and removing hydrogen ions. Note that the glutamate comes
mainly from the liver, which thus participates indirectly in acid-base
balance.
Note that for the reaction:
NH3 + H+ <==> NH4+
the pK' is 9.0, in other words, only at a pH of 9.0 will equal concentrations
of ammonia and ammonium be present at equilibrium.
The lower the pH, the higher the concentration of ammonium ion relative to
that of ammonia. This is a wonderful mechanism for concentrating ammonium
ion in the urine - the renal cells make ammonia which diffuses into the urine,
hydrogen ions in the urine
bind the ammonia to make ammonium, and more ammonia can therefore diffuse
passively across the renal cells into the urine, to be snapped up in turn!
As they often do, physiologists have complicated things by labelling this
simple process - they call it "nonionic diffusion".
How can we determine the amount of ammonium ion in urine?
Ammonium concentration can be directly determined in the laboratory,
but a convenient 'trick' is to
estimate it by calculating the urine net charge, often referred to
rather less accurately as the "urinary anion gap". This is:
Urine net charge = [Na+]U + [K+]U - [Cl-]U
It is easy to see how if there is a high concentration of ammonium in the
urine, the chloride concentration will usually be far higher than the
sum of the Na and K concentrations - the extra chloride goes into balancing
the positive charge provided by the NH4+. Thus, with lots of
ammonium, the urine net charge will be negative - this is the
normal compensation for metabolic acidosis.
Causes of Hyperchloraemic Acidosis
Acidosis of this type is often both initiated and perpetuated by renal
tubular dysfunction, hence the term "renal tubular acidosis" (RTA). There are
however other causes of "normal anion gap acidosis".
If you look at the standard textbooks, you'll probably see something
resembling the following list:
("Type IV RTA")
( with mineralocorticoid deficiency/resistance,
tubulointerstitial disease, and a variety of drugs
).
Other renal:
- Carbonic anhydrase inhibitors (resembles Type II RTA)
- Primary hyperparathyroidism (resembles Type II RTA)
- With renal dysfunction, and hyperkalaemia related to
pentamidine,
non-steroidal anti-inflammatories, cyclosporine.
Gastrointestinal bicarbonate loss:
related to diarrhoea,
or pancreatic loss of bicarbonate;
Ureterosigmoidostomy
Administration of the following:
- Large amounts of phosphate
- Cation exchange resins
- Calcium chloride (!)
- Ammonium chloride, hippurate
- Parenteral hyperalimentation
(Chloride salts of basic amino acids
(lysine, arginine) and insufficient alkali)
- Large amounts of NaCl (saline: "expansion acidosis")
Note that in the above list, most of the non-renal causes are self evident,
and would have to be persistent, or otherwise a normally functioning kidney
would compensate for the acidosis. We will therefore concentrate on renal
causes, so called "Renal Tubular Acidosis".
RTA excludes people with severe renal dysfunction, where acidosis
is often associated with a moderately raised anion gap, and is due to
overall renal dysfunction with inability to clear hydrogen ions.
Regarding RTA, different authors
have radically different classifications, and some have even tried to
abandon the concept of "proximal" and "distal" RTA. This latter approach
does not at present seem to have much utility, and we have thus used the
more traditional approach. Also note that "Type III" RTA doesn't exist -
previously, people used it to (for example) categorise cases where there seemed to
be a mixture of type I and type II.
Proximal RTA
(Type II RTA)
This disorder often appears milder in its presentation, but is more
difficult to treat than distal RTA. This is because the cause is
proximal tubular wasting of bicarbonate - diminished bicarbonate
resorption results in loss of bicarbonate in the urine, until the
serum bicarbonate level drops to a level where equilibrium is reached
(usually at about 15-17 mmol/l). Whenever the serum level rises, the
more bicarbonate is simply lost in the urine. Many cases of type II RTA
are associated with generalised proximal tubular dysfunction, presenting
as the "Fanconi syndrome" (proximal RTA together with proximal tubular
loss of glucose, amino acids and phosphate). Presentation may be with
features of the underlying disease, or with features of acidaemia and
hypokalaemia. Bone disease is fairly common.
We can conveniently classify causes as follows:
Causes of Proximal RTA:
- With generalised proximal tubular dysfunction
- Genetic diseases (a vast number: tyrosinaemia,
cystinosis, medullary cystic disease, fructose intolerance,
glycogen storage disease type I, Wilson's disease, Lowe
syndrome, galactosaemia..), as well as isolated primary
proximal RTAs;
- Dysproteinaemias
- vitamin-D deficiency/resistance, ? hypocalcaemia!
- Sjogren's syndrome
- Transplant rejection
- Drugs & toxins: expired tetracycline, streptozotocin,
cyclosporine, gentamicin, coumarin, lead, cadmium, Hg,
- Isolated proximal RTA (uncommon)
- Hereditary
- carbonic anhydrase II deficiency (+- osteopetrosis)
- Similar picture seen with carbonic anhydrase inhibitors
Treatment of Proximal RTA
This is difficult. Orally administered alkali is simply lost in the urine.
Always remember to identify and treat (if possible) the underlying cause!
One management strategy is to restrict sodium intake (and possibly
even administer a thiazide diuretic!) This resets glomerulotubular balance,
with increased proximal tubular resorption of sodium and bicarbonate, and
less bicarbonate wasting.
Distal RTA
(Type I RTA)
This is often a far more serious disorder than type II RTA, not only with
acidosis and hypokalaemia, but also with frequent nephrolithiasis and
progression to nephrocalcinosis in a large proportion of cases. The
response to treatment with oral alkali is however good.
Causes of distal RTA
There are several distinct types of distal RTA:
- Classical "gradient-limited" RTA type I
Here it is thought that hydrogen ions leak back into the
interstitium, despite adequate secretion. Serum potassium levels
are low. (Some have claimed that those with low potassium levels
have a defect in the H+/K+ antiporter, rather than inability
to maintain a hydrogen ion gradient).
The disorder may occur:
- As 'Primary'(sporadic or hereditary) RTA,
with no other associated disease;
- Associated with a variety of genetic diseases
(Ehlers-Danlos syndrome, Marfan's, sickle-cell disease,
ellliptocytosis, medullary cystic disease, Fabry's,
Wilson's)
- With 'autoimmune disorders' (hypergammaglobulinaemia,
Sjogren's, systemic lupus, rheumatoid arthritis, vasculitis,
chronic active hepatitis, cirrhosis including primary
biliary cirrhosis, thyroiditis)
- With hyperparathyroidism, vitamin D intoxication, and
other disorders of calcium metabolism;
- With chronic pyelonephritis, renal transplantation,
amyloidosis
- With drugs such as analgesics, vanadate and toluene;
- In some cases, the actual proton pump in the distal tubule
may be defective. This has been termed "rate limited secretory
distal RTA".
- Voltage-dependent RTA
Distal tubular sodium resorption is impaired.
This diminishes the size of the lumen-negative voltage across
cells lining the tubule, and it is this voltage change that
impairs secretion of both hydrogen ions and potassium, resulting
in renal tubular acidosis and hyperkalaemia. Often there
is an underlying cause of the impaired sodium resorption, for
example amiloride, triamterene or even lithium administration
(which may cause a combined voltage and H+/K+ pump problem).
- Ammonium ion synthesis/transfer defects
Low glomerular filtration states, defects in medullary recycling
of ammonia (!), and even severe hyperkalaemia have been implicated
in this rather speculative disorder. Interstitial nephropathy
may also lead to this problem.
- Amphotericin B induces an interesting permeability defect
in the distal tubule - protons appear to leak back from the tubule
into cells, but some have questioned this theory, suggesting that
H2CO3 leaks back, or even that bicarbonate leaks out!
Treatment of distal RTA
The mainstay of treatment is provision of oral alkali, for example
Shohl's solution (sodium citrate + citric acid), in a dose of 1-4 mmol/kg/day
(in an adult, about 30ml QID is often sufficient).
Hypoaldosteronism & RTA
(Type IV RTA)
This disorder has a number of causes, including:
- Tubulointerstitial disease (a form of resistance to aldosterone)
- Aldosterone deficiency (with or without glucocorticoid deficiency,
i.e. Addison's disease)
- Deficient renin secretion
- Resistance to aldosterone:
- hereditary
- Pseudohypoaldosteronism (Types I, II)
- Drugs: amiloride, triamterene, spironolactone,
trimethoprim, ACE inhibitors, long-term heparin administration!
Pathogenesis is probably complex, including decreased stimulation of proton
secretion due to the absence of aldosterone effect, a mild voltage gradient
defect, and hyperkalaemia (which interferes with ammonia production and
transport). Urinary acidification is normal (pH can drop below 5).
Treatment of RTA consequent on hypoaldosteronism
Again, identifying and treating the cause is important.
Management of the hyperkalaemia is vital, and will often result in
resolution of the acidosis. Select cases may benefit from mineralocorticoid
administration, or even use of loop diuretics (furosemide).
Distinguishing between RTA Types
-
First we have to establish whether RTA is present. If the patient has
hyperchloraemic metabolic acidosis, then one should check the urinary
pH, and more importantly, the urine net charge.
If the urine net charge is negative,
then ammonium production is appropriately high, and RTA
types I and IV are unlikely. It is possible that the patient
has proximal RTA, and if other causes of hyperchloraemic acidosis with
a negative urine net charge are not present (intestinal bicarbonate loss,
administration of acidic salts or ammonium chloride, use of acetazolamide)
then the diagnosis of proximal RTA can be made by administering bicarbonate,
and calculating the fractional excretion of filtered bicarbonate. If this
is over 15%, the diagnosis is made. (Speak to your laboratory about
determining the fractional excretion of filtered bicarbonate). Other
clues to the presence of proximal RTA might be other features of the
Fanconi syndrome. Alternatively, one could try administration of Shohl's
solution, and if the response is poor, strongly suspect type II RTA!
- If urine net charge is positive, types I or IV RTA are likely.
Check the serum potassium. Secretory distal RTA will be associated
with hypokalaemia (or sometimes, normokalaemia), and
respond well to administration of alkali (e.g. Shohl's solution).
Note that with classical type I RTA (gradient-
limited secretory distal RTA) the urine pH will tend to be over 5.5,
while with rate-limited secretory distal RTA, pH is often below 5.5.
Be cautious in using urinary pH - urea-splitting organisms present in
urine might raise pH, and any cause of volume or potassium depletion
will also raise it remarkably).
One problem that can be confused with rate-limited secretory distal RTA
is defective NH3 production. These can be distinguished by giving bicarbonate
and checking the urinary PCO2. If there is a distal defect in hydrogen
ion secretion, the urine PCO2 minus the blood PCO2 will be abnormally
low (under 3.3kPA). Normally, alkalinisation of the urine to a pH of
over 7.0 by NaHCO3 administration results in bicarbonaturia, and this
bicarbonate binds distally secreted hydrogen ions to form H2CO3. The H2CO3
in turn breaks down to form CO2 and H2O, resulting in a urinary PCO2
of over 3.3kPa.
Amphotericin B toxicity may resemble gradient-limited secretory
distal RTA, but here again, urine PCO2 - blood PCO2 will be normal,
in contrast to the low value found in gradient-limited secretory distal RTA.
If you are still unsure about the presence or absence of
distal RTA, ammonium chloride loading has been used to distinguish between
distal RTA (where urine pH fails to drop below 5.5) on the one hand,
and proximal or type IV RTA on the other (where the pH drops). In rate-
limited distal RTA, the pH should also drop. Avoid this test.
- If there is hyperkalaemia, suspect type IV RTA, or voltage-distal RTA.
In voltage distal RTA, the urine pH tends to be over 5.5, while with
hypoaldosteronism, the pH is often under 5.5.
It may be necessary to determine serum aldosterone levels (low with aldosterone
deficiency, high-ish with resistance), although often the clinical context
will tell you what to expect.
References
- Smulders et al, Arch Intern Med 1996 156 pp1629-36.
Renal Tubular Acidosis. Pathophysiology and Diagnosis.
A complex but fairly good review.
- Kokko JP. (Chapter 75) and Chesney RW (Chapter 82) in
Bennett & Plum. Cecil Textbook of Medicine, 20ed. Pp 546-7,
594-8.
Good overall reviews of renal tubular acidosis. Better, in
our opinion, than Harrison's.
- DuBose TD (Chapter 50) and Asplin JR & Coe FL (Chapter 278)
in Fauci AS et al.
Harrison's Principles of Internal Medicine. 14ed. pp 279-282, 1566-8.
Reasonable stuff on RTA.
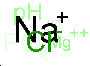 Back Up
Back Up
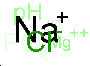 Back Up
Back Up